

EKOS
Control Unit 4.0
Since its inception, EKOS®, a BTG International group company, has one goal in mind: to develop life-enhancing and lifesaving treatments for vascular thrombosis (blood clots) that occur in arteries and veins. In 2005, EKOS pioneered the development and clinical application of ultrasound infusion technologies in medicine by introducing its first system to treat blood clots.
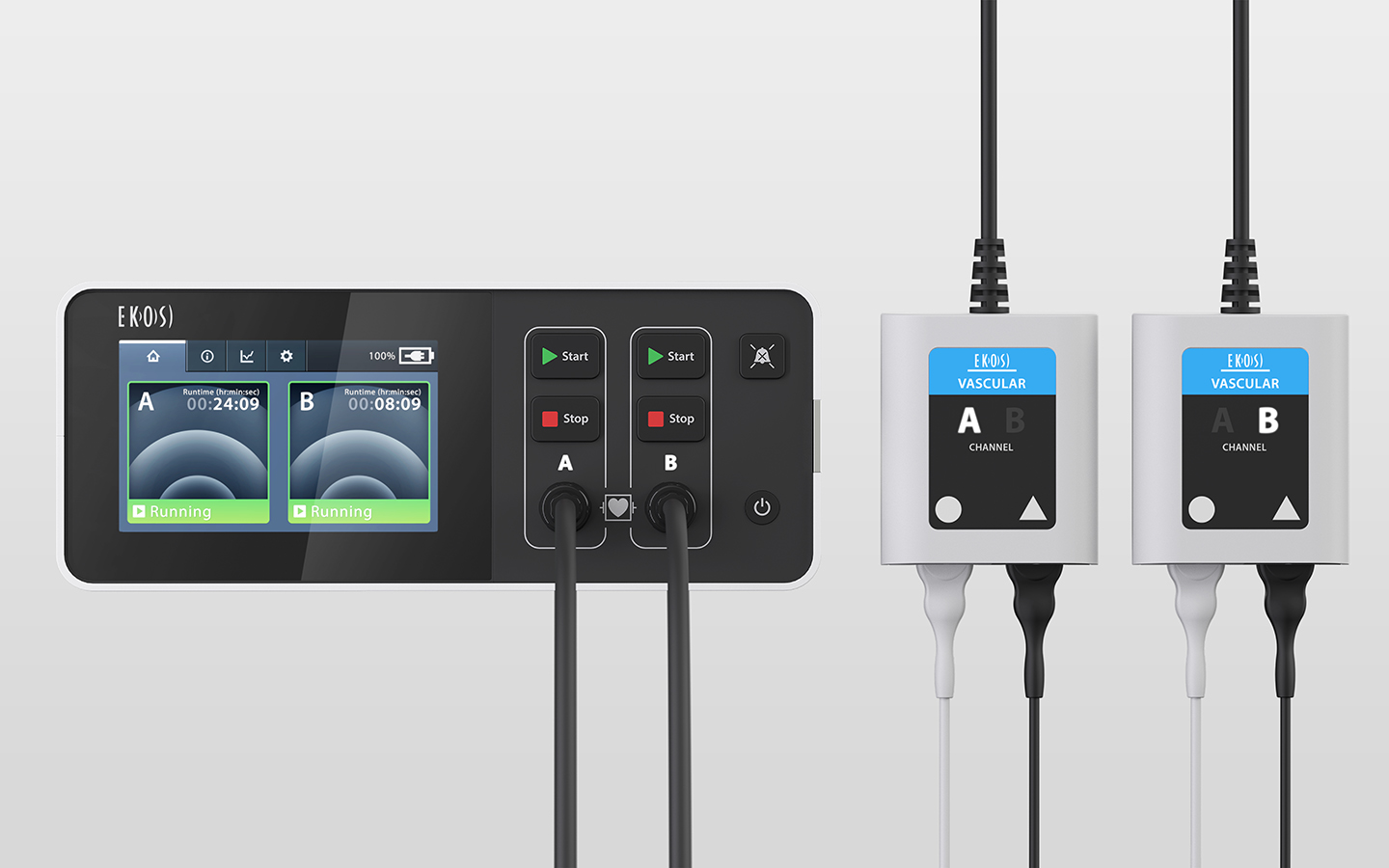
Looking to refresh their pulmonary embolism (PE) treatment device, EKOS enlisted Tactile to redesign the control unit that powers the EKOS System, the user interface, and the system’s packaging.

Simultaneous Flow
Designing medical devices has become more than an interest for us—it’s a passion. Tactile’s main challenge was to redesign the original Ekosonic Endovascular system to minimize the overall product footprint while adding functionality to treat two pulmonary arteries simultaneously. The team minimized the control unit complexity from an ID and UX perspective to streamline the overall set-up procedure and help monitor the treatment.
Currently the only FDA-approved device to treat PE on the market, the EKOS Control Unit 4.0 allows physicians to easily monitor and independently manage two EKOS devices simultaneously, simplifying bilateral PE treatment. The EKOS Control Unit 4.0 provides the power to the EKOS System’s ultrasonic core device that uses acoustic pulses to efficiently and safely dissolve blood clots and restore blood flow in patients with PE.
Reducing
Anxiety
Along with designing the control unit, Tactile developed an intuitive, highly visible user interface that reduces alarm fatigue and brings critical information to the forefront. Several iterations of the interface design proved to be pivotal in understanding how doctors and technicians interact with the interface and the patient’s response to the graphics and visuals.


Visceral Packaging
EKOS wanted to streamline the EKOS Control Unit 4.0 packaging to improve efficiency when identifying the products on shelves. Tactile designed the infusion catheter/ultrasonic core packaging and the console box package.
The newly designed packaging simplifies shelf display for quick, intuitive identification of each product component.
